AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
As element metal or nonmetal4/5/2023  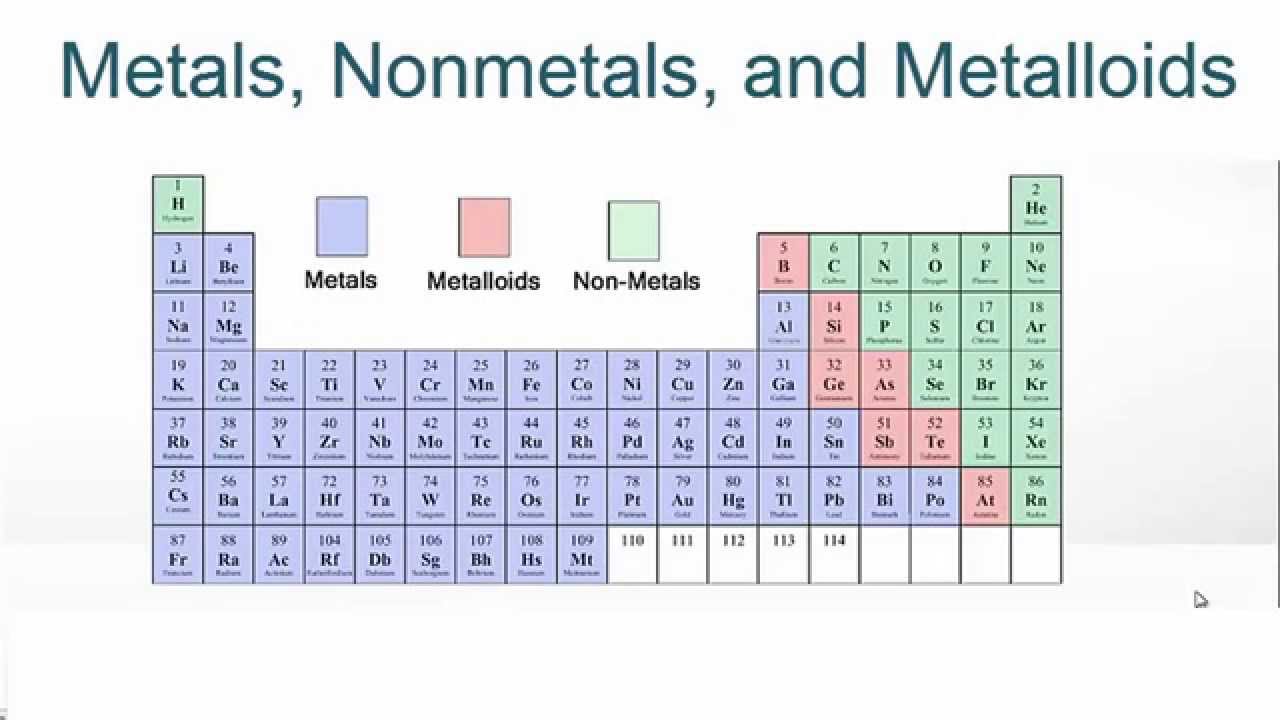
Nonmetals that form giant structures, such as chains of up to 1,000 atoms (e.g. oxygen, sulfur and bromine) have low melting and boiling points as they are held together by weak London dispersion forces acting between their atoms or molecules. The internal structures and bonding arrangements of the nonmetals explain their differences in form. The solid elements have low densities, are brittle or crumbly with low mechanical and structural strength, and poor to good conductors. The fluid nonmetals have very low densities, melting points and boiling points, and are poor conductors of heat and electricity. Bromine, the only liquid, is so volatile that it is usually topped by a layer of its fumes sulfur is the only colored solid nonmetal. General properties Physical Physical properties apply to elements in their most stable forms in ambient conditionsĪbout half of nonmetallic elements are gases most of the rest are shiny solids. The superheavy elements copernicium ( Z = 112), flerovium (114), and oganesson (118) may turn out to be nonmetals their status has not been confirmed. 
Astatine, the fifth halogen, is often ignored on account of its rarity and intense radioactivity theory and experimental evidence suggest it is a metal. The status of a few elements is less certain. Of the 118 known elements no more than about 20% are regarded as nonmetals. At other times they are counted as nonmetals in light of their nonmetallic chemistry. The elements commonly recognized as metalloids namely boron silicon and germanium arsenic and antimony and tellurium are sometimes counted as an intermediate class between the metals and the nonmetals when the criteria used to distinguish between metals and nonmetals are inconclusive. The authors recognized carbon, phosphorus and selenium as nonmetals Vernon had earlier reported that these three elements were instead sometimes counted as metalloids. Fourteen almost always recognized are hydrogen, oxygen, nitrogen, and sulfur the highly reactive halogens fluorine, chlorine, bromine, and iodine and the noble gases helium, neon, argon, krypton, xenon, and radon (see e.g. Īlthough Steudel, in 2020, recognised twenty-three elements as nonmetals, any such list is open to challenge. The decisions involved depend on which property or properties are regarded as most indicative of nonmetallic or metallic character. 
Since there is no rigorous definition of a nonmetal, some variation exists among sources as to which elements are classified as such. Fourteen elements are always recognized as nonmetals and nine other elements partially qualify as nonmetals.ĭefinition and applicable elements Ī nonmetal is a chemical element deemed to lack a preponderance of metallic properties such as luster, deformability, good thermal and electrical conductivity and the capacity to form a basic (rather than acidic) oxide. Some elements have a marked mixture of metallic and nonmetallic properties, which of these borderline cases are counted as nonmetals varies on the classification criteria. While the term non-metallic dates from as far back as 1566, there is no widely agreed precise definition of a nonmetal. Nearly all nonmetals have individual uses in medicine, pharmaceuticals, lighting, lasers, and household items. Living organisms are composed almost entirely of the nonmetals hydrogen, oxygen, carbon, and nitrogen. Most nonmetals have biological, technological or domestic applications. 
Five nonmetallic elements, hydrogen, carbon, nitrogen, oxygen and silicon, largely make up the Earth's crust, atmosphere, oceans and biosphere. Two nonmetals, hydrogen and helium, make up about 99 percent of ordinary matter in the observable universe by mass. Nonmetal atoms tend to attract electrons in chemical reactions and to form acidic compounds. The electrons in metals are generally free-moving and this is why metals are good conductors and most are easily flattened into sheets and drawn into wires. The electrons in nonmetals behave differently from those in metals with some exceptions, the electrons in nonmetals are fixed in place, resulting in nonmetals usually being poor conductors of heat and electricity, and brittle or crumbly when solid. In chemistry, a nonmetal (or non-metal) is a chemical element that generally lacks a predominance of metallic properties they range from colorless gases (like hydrogen) to shiny solids (like carbon, as graphite). Hydrogen is usually in group 1 (per the below full table) but can be in group 17 (per the above extract). There is no precise definition of a nonmetal which elements are counted as such varies. Extract of periodic table showing how often each element is classified as a nonmetal:ġ4 effectively always 3 frequently 6 sometimes ( metalloids) 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
